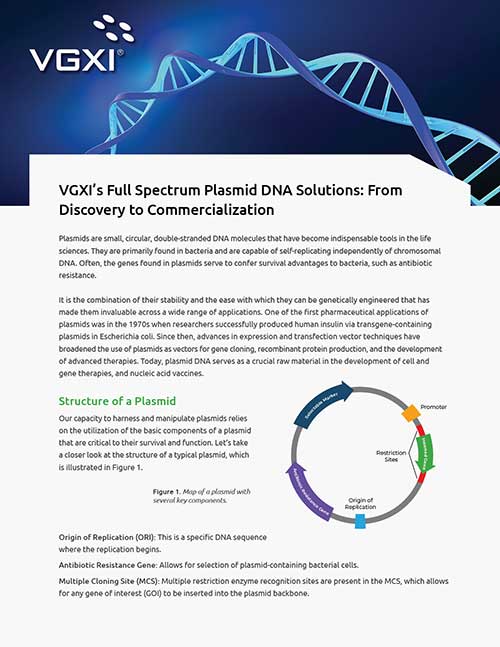
VGXI Resources
Welcome to our Plasmid DNA information hub! This is your one-stop destination for the latest pDNA news and events, comprehensive FAQs, and access to our knowledge center and blog. Discover the expertise and innovations that make VGXI’s Plasmid DNA manufacturing and purification a leading-edge CDMO in the industry.