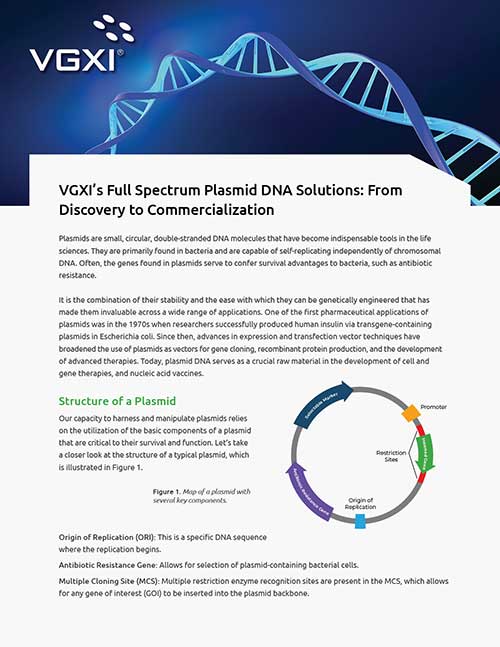
Technical Expertise
Leading-edge CDMO
Since 2003, VGXI has solidified its position as a leading-edge Plasmid DNA CDMO, specializing in the production of high-quality plasmid DNA products to support the advancement of innovative cancer therapies and DNA vaccines that address some of the most pressing challenges in modern medicine. Notably, VGXI has played a pivotal role in manufacturing pDNA in response to critical emerging diseases, such as Ebola, MERS, Zika, and the recent COVID-19 pandemic.
Our industry-leading Plasmid DNA expertise coupled with our patented pDNA manufacturing process and state-of-the-art facilities, enables VGXI to deliver tailor-made manufacturing solutions to meet the highest quality and regulatory standards in the industry for every program. We have a reputation of success in manufacturing challenging DNA products under cGMP conditions with exceptional yield, quality, and prompt delivery for clinical trials in the US, EU, Asia, Canada, and Australia. Our products have passed rigorous reviews by several international regulatory agencies.
From small-scale to commercial production, VGXI’s client-focused Plasmid DNA manufacturing team will build a solid foundation for your program with our proven DNA plasmid production services. We manufacture quality DNA plasmids across a range of service levels:
- Pre-clinical Plasmids Production Service provides high purity plasmid preparations for early research, in vivo animal studies, and preclinical assessments for advanced therapies.
- Highly Documented (HD) Plasmid Production Service provides high supercoil, low endotoxin plasmids for use as an ancillary raw material in cGMP manufacturing and IND enabling studies.
- cGMP Plasmid Production Service produces injectable-grade plasmid DNA for human clinical trials, diagnostic reagents, and the GMP production of cell and gene therapies.
Our end-to-end solutions include Plasmid DNA Process Development & Optimization, Tech Transfer Support, GMP Cell Banking & Testing, Fill/Finish Services, Drug Product Stability Testing, and Rapid TAT Manufacturing.
VGXI’s ability to work with unique requirements and create custom manufacturing solutions is based on the unsurpassed knowledge, expertise, and ingenuity of our team comprised of leading experts in Process Development, Quality Control, Quality Assurance and Validation of plasmid-based biopharmaceuticals.